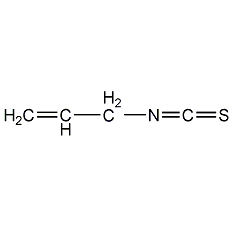
Structural formula
| Business number | 018A |
|---|---|
| Molecular formula | C8H15NO3 |
| Molecular weight | 173.21 |
| label |
6-Aminocaproic acid |
Numbering system
CAS number:57-08-9
MDL number:MFCD00004424
EINECS number:200-310-8
RTECS number:None
BRN number:None
PubChem ID:None
Physical property data
1. Characteristics: white crystal .
2. Density (g/mL,25/4℃) : Undetermined
3. Relative vapor density (g/mL,Air=1): Undetermined
4. Melting point (ºC): 104~105.5
5. Boiling point (ºC,Normal pressure): Undetermined
6. Boiling point (ºC,5.2kPa): Undetermined
7. Refractive index: not OK
8. Flashpoint (ºC): Undetermined
9. Specific optical rotation ( º): Undetermined
10. Spontaneous ignition point or ignition Combustion temperature (ºC): not OK
11. Vapor Pressure (kPa,25ºC): Undetermined
12. Saturated vapor pressure ( kPa,60ºC): Undetermined
13. Heat of combustion (KJ/mol): Undetermined
14. Critical temperature (ºC): Undetermined
15. Critical Pressure (KPa): Undetermined
16. Oil and water (octanol /Water) partition coefficient pair Value: Undetermined
17. Explosion upper limit (%,V/V): not OK
18. Lower explosion limit (%,V/V): not OK
19. Solubility: Not available Sure.
Toxicological data
None
Ecological data
None
Molecular structure data
5. Molecular property data:
1. Molar refractive index: 44.30
2. Molar volume (m3/mol):161.1
3. isotonic specific volume (90.2K):405.2
4. Surface tension (dyne/cm):39.9
5. Polarizability(10-24cm3):17.56
Compute chemical data
1. Reference value for hydrophobic parameter calculation (XlogP): 0.1
2. Number of hydrogen bond donors: 2
3. Number of hydrogen bond acceptors: 3
4. Number of rotatable chemical bonds: 6
5. Number of tautomers: 2
6. Topological molecular polar surface area (TPSA):66.4
7. Number of heavy atoms: 12
8. Surface charge: 0
9. Complexity: 156
10. Number of isotope atoms: 0
11. Determine the number of atomic stereocenters: 0
12. Uncertain number of stereocenters: 0
13. Determine the number of stereocenters of chemical bonds: 0
14. Uncertain number of chemical bond stereocenters: 0
15. Number of covalent bond units: 1
Properties and stability
None
Storage method
This product should be kept sealed.
Synthesis method
None
Purpose
Pharmaceutical intermediates.
extended-reading:https://www.cyclohexylamine.net/low-odor-catalyst-pt302-dabco-hard-foam-catalyst/extended-reading:https://www.bdmaee.net/potassium-acetate/extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/bismuth-neodecanoate-CAS34364-26-6-bismuth-neodecanoate.pdfextended-reading:https://www.bdmaee.net/catalyst-9727/extended-reading:https://www.bdmaee.net/wp-content/uploads/2022/08/-B-16-amine-catalyst-B16–B16.pdfextended-reading:https://www.newtopchem.com/archives/1822extended-reading:https://www.newtopchem.com/archives/1141extended-reading:https://www.cyclohexylamine.net/addocat-106-teda-l33b-dabco-polycat/extended-reading:https://www.newtopchem.com/archives/40504extended-reading:https://www.cyclohexylamine.net/polyurethane-catalyst-dabco-dc2-strong-gel-catalyst-dabco-dc2/